I was asked a question about electron orbitals via an email the other day – which basically asked do electrons orbit the atom or not? It is an interesting question but if you only have a few seconds the answer is no.
One thing we know is that when an object has an orbital path, this object is undergoing constant acceleration – remember acceleration is a change in velocity which as reference to direction. Direction must be changing if you are spinning round and round in a circle. Electromagnetic radiation is a complex thing, but one thing I can tell you about it is that if a charged particle is undergoing acceleration like this will emit electromagnetic radiation. This concept is a little tricky – but try to appreciate that because an electron has an electric charge it is associated with an electromagnetic field. These electromagnetic fields are fundamental ideas in particle physics – when we have a positive charge it creates a distortion such that if a negative charge is placed near it a force will act upon it. Now if we have an electron accelerating this electron can and will produce excitation in the electromagnetic field – and indeed what is electromagnetic radiation but excitation in the electromagnetic field. So if our electron was to emit electromagnetic radiation, the electron would lose energy which would mean it would collapse inwards to the nucleus. This does not happen: conclusion, the electron is not orbiting.
The orbiting electron is the Niels Bohr model, which is fine and works to help elementary physicists understand the electron but if you are going to break into the realms of quantum mechanics then I am afraid you need an all new picture – you need to adopt the Schrodinger model which I show you below.
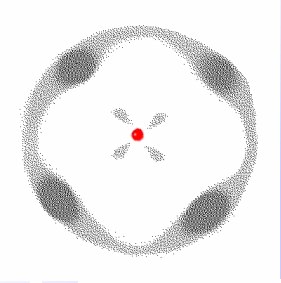
This is where the phrase “quantum fuzz” comes from from and embodies some of the important points of quantum mechanics. Most fundamental is the fact that I cannot tell you where an electron will be next given a set of initial conditions, and even more disturbingly – I cannot measure the position and the velocity of the electron to a good level of precision (this is Heisenberg’s uncertainty principle). So what I am left with is a set of probabilities of where I will find the electron which is what you are seeing above – the denser areas being the more probable but none the less there are a huge range of possible locations for the electron – and if an electron can go somewhere it will, some places just take longer than others to reach.
This set of probabilities is what we were alluding to when we were talking about electron shells. It is the mathematics and physics behind this which explains why electrons are arranged in “shells”, why there are a certain number of different quantum states within a given energy level and why the orbitals have the shape they do. So perhaps by calling them orbitals, we are falling on our own sword?

By the same reasoning an electron must accelerate in order to remain within the denser areas of the orbital or, perhaps more accurately (I think), the shape of the orbital implies the electrons are usually accelerating. If so, what gives them the energy to do this?
LikeLiked by 1 person
I understand why you would think that because I did too for ages – but I think you have to start from the point that there is no way an electron can be accelerating – because accelerating through a charged fields needs energy, which means the electron would be ontop of the nucleus in seconds – and given the majority of matter is space between “orbitals” and the nucleus, that would be the end.
This is really the heart of quantum mechanics – and why people say if you’re comfortable with quantum mechanics you simply haven’t understood it.
There always has been a big problem with thinking of electrons as points orbiting a bigger point. The electrons are actually a cloud of probability that shrouds the nucleus. So in a sense they are the strings of an instrument, oscillating at agreeable frequencies, not deviating unless excited, and not accelerating.
LikeLiked by 1 person
I never thought through that commonly used phrase about electrons orbiting the nucleus before. This is really interesting.
LikeLiked by 1 person
Indeed it is one of those things that when you actually give it some thought really is quite amazing
LikeLike
I see your website is similar to my page. Do you allow guest posts?
I can write unique content for you. Let me know if you are interested.
LikeLike
Hi, sorry we don’t currently accept guest posts unless the page has significant traffic. Thanks.
LikeLike
I want to understand how do we use probability in finding where the electron will be? What is the uncertainty?
LikeLike
Pingback: The misleading evidence that fooled scientists for decades - Crypto Commune·
Pingback: The misleading evidence that fooled scientists for decades - StuntFM 97.3·
Pingback: The misleading evidence – befarmed.wordpress.com·
Pingback: 4 convincing scientific theories that fooled scientists for decades | Technology News and Markets·