Another little piece of chemistry today, because we did say we would try to keep a broad mind. It is often of bemusement to me why organic chemistry takes up so much less importance than inorganic; organic chemistry is naturally fascinating due to its close link with the human experience, as well as covering 90% of chemical compounds. I was actually browsing a textbook that stated organic chemistry is often overlooked – whilst giving 8% of the pages in this generalist chemistry book to the subject. I did email and enquire, but the question was greeted with no great enthusiasm.
Having said that, there is one very good reason why organic chemistry need not cover 50% of the generalists paper which is the functional approach. With inorganic chemistry one must understand quite a lot about the periodic table, molecular structures, chemical bonding etc. to predict quite basic reactions. Organic chemistry is much neater than this. The size of the molecules in organic chemistry can be a little daunting; take for example triacylglycerol molecules (basically margarine) – this has a core of glycerol and then three tails as shown in this diagram (the glycerol core is shown on the left, then the full triacylglycerol molecule on the right).
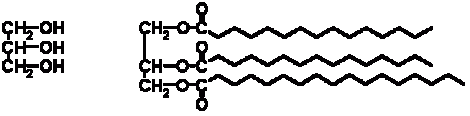
These “tails” represented by the wavy lines can be in the 10’s of thousands of carbon and hydrogen atom chains, so how does one make sense of it all? And this is where the functional approach comes into organic chemistry which is quite beautiful. If says that no matter how big and complex a molecule may look we can identify functional groups within it, which we will define shortly. We can split the molecule into functional groups and “the rest”. When we are considering what will happen in a reaction with the molecule we can ignore the rest (more or less) and only consider the impact on our functional groups. As a result, that beastly looking triacylglycerol is rationalised into three identical functional groups – plus the rest. Sounds a bit better right? Here are some of the functional groups that are commonly used in organic chemistry.
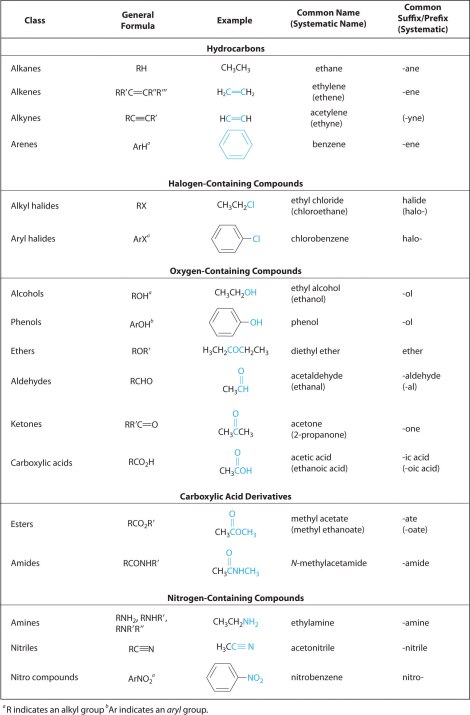
Some of these look really complex by they really are not. Let us take an example of acetic acid reacting with methanol. This is what you have:
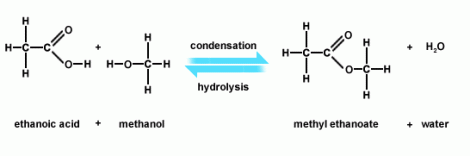
Now if you look at the ethanoic acid you should be able to identify the section of the molecule that has the COOH on the right. This makes this a carboxylic acid – this is its functional group. On the methanol, you should be able to recognize the OH group. This is what makes this an alcohol. Remember what we said – identify the functional groups and the rest is totally irrelevant. So all that is going to happen is that the alcohol section and the carboxylic acid section will interact – and ehat happens is I loose my hydrogen from my ethnoic acid and my alcohol group OH from my methanol to give me methyl ethanoate. Now you should notice, but I will forgive you if you didn’t, that I have a O=C-O bonding in there, which now makes the functional group of the product an esther – with water formed from the loose parts that have been stripped out. This might all seem very complex – but actually the beauty is I can generalise and take ANY carboxylic acid, and ANY alcohol, react them in this way and fuse them together with water given of.
The diagram above identifies that this is a condensation reaction – and the reverse of this reaction would be hydrolysis (the addition of water to a larger molecule to make two smaller molecules). What you should hopefully be able to appreciate now without a huge conceptual leap is that I can write out a general formula for any reaction of this type and it will always look the same. So what I end up being able to do is identify functional groups within molecules so I know exactly what will react, and generalise exactly how these parts will react. I have drawn out some reactions where I have used R to represent the inert part of the molecule that we are not interested in:
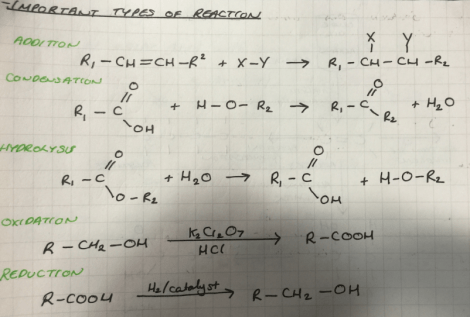 Where I have written some substances on top of the arrows these are general reagents/catalysts that are typically used in the reactions. So this is really quite neat. If I have an alkene functional group, for example which I would identify with the CH=CH bonding and I have say Fl2 gas – it isn’t a great leap for me to say that this is an addition reaction, so if you look above I have my alkene and I have my flourine – and the double bonds will break and form a single bond with the carbon atoms. Then I am now in the functional group of the haloalkenes, where substance like chloroform belong.
Where I have written some substances on top of the arrows these are general reagents/catalysts that are typically used in the reactions. So this is really quite neat. If I have an alkene functional group, for example which I would identify with the CH=CH bonding and I have say Fl2 gas – it isn’t a great leap for me to say that this is an addition reaction, so if you look above I have my alkene and I have my flourine – and the double bonds will break and form a single bond with the carbon atoms. Then I am now in the functional group of the haloalkenes, where substance like chloroform belong.
This approach has far reaching consequences – for example each protein is made up of amino acids – which must have a carboxylic acid functional group and an amino functional group. Then what happens to build proteins is that these amino acids join up in addition reactions with themselves – leaving a carboxylic acid functional group on one end, an amino group on the other end, then amide connections with the amino acid residues. It really is quite a fascinating approach that has far reaching consequences into everything organic – humans, food, pharmaceuticals and plants (cellulose is for example a polysaccharide which has alcohol and hemiacetal functional groups).
Hopefully that has been interesting – I appreciate that it has been a bit more technical than usual but do ask if you have any questions. I am going to post a few softer posts soon too!

I ma not that good in chemistry but Very interesting, looking forward to your next post
LikeLiked by 2 people
I think it is hard for any of us to feel good at Chemistry it is such a pain! Thank you for visiting 🙂
LikeLiked by 1 person
thank you for post
LikeLike
erm …..well yes …I can see it must be interesting ..chemical building blocks …reactions etc …but BUT my head hurts …still that is to be expected …enjoying the ‘Elegant Universe’ tho …I think someone with a brain far greater than mine needs to invent more numbers …it’s about time we brought ‘clocks’ into the 21st century …but I’m digressing:D:D:D
LikeLike
I’m here for the hors d’oeuvres and to mingle, oooh hey now, there’s Pat, excuse me ….. …. *wink*
LikeLiked by 1 person
I love organic chemistry!
LikeLiked by 1 person