For anyone who regularly follows my blog, or for those I interact with regularly I must apologise – if you follow my twitter you will know I got marooned on the small island of Jersey this week which through everything off kilter – my studies, my running, my blog and my German. Schade! Anyhow, I am back in the comfort of my small little flat in London now, having done the weeks washing and caught up with my degree work so let’s get going again.
I am currently fulfilling the Chemistry requirements of my generalist science section, which I do find fairly enjoyable. I have to say however there is an inner frustration you have to wrangle with when studying Chemistry because there are large patches of it for which we don’t know why it happens. We have the rules very well pinned down and we can say that action A happens because of rule B, however why rule B should even be a rule in the first place is sometimes, but not always ambiguous. That said – it is a great subject and not one that should be overlooked by scientists in any discipline.
I imagine there are going to be quite a number of posts from me on Chemistry related themes over the next few weeks, given the studies I am doing, but for today I thought it would be good to introduce some of the basic concepts of the periodic table. I am not going to be going to go into basic detail on the periodic table – I assume a very simple working knowledge but if you do find any of it confusing, just ask.
Group 1 metals
Anyone who took Chemistry at GCSE level will probably remember playing with group one metals, dropping them into water and observing reactions similar to these (the less extreme ones anyway).
Group one metals are interesting metals indeed – they are soft metals that quickly go dull when left to react with Oxygen. The most interesting thing about them is the explosive reaction they have when they react with water and the reason for this is very simple. Take a look at the electron configuration of sodium:
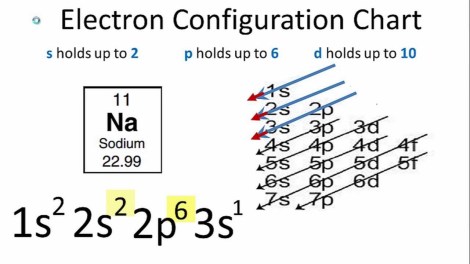
Electron configuration of sodium
All group one metals will react with chlorine to form white solid compounds of the format one group one metal for every one chlorine atom (this is just an example of one of many chemical reactions)
Examples are sodium, potassium and francium.
Group 2 metals
The alkaline earth metals exhibit very similar properties – except rather than having one electron in the valence shell they have two. This of course means that they have lower reactivity, although they still are highly reactive. The group two metals would react with things like oxygen with one group 2 metal for every 2 oxygen.
All group two metals will also react with chlorine – but since the valency of chlorine is unchanged, but our group 2 metals need to shed two electrons we are left with two chlorine atoms for every one group two metal. Take a look at the reaction in the Lewis diagram structure.

Lewis diagram for the reaction of Magnesium and Chlorine
Transition metals
The transition metals go from group 3 through to group 12 and contain 38 metals. In general there is less to say about these – that isn’t necessarily because the chemistry is any less rich or interesting it is more a result of the fact that the rules are much less prevalent – I can’t give the same quick rules and characteristics like I can for group one or two metals. There are some things that are generally true of the transition metals however;
- They are malleable and ductile;
- They are conduct both heat and electricity; and
- The electrons they use to bind with other atoms are found in more than one subshell.
It is this last point which is why they don’t observe such screamingly obvious logic, but if you think about they way electron subshells are filled, you were bound to get to scenarios where this happened. Iron, cobalt and nickel are the standout transition metals, since they are the only metals known to produce a magnetic field.
Group 13
This is the Boron family, which tend to favour +3 oxidation states – they need to shed three of their outer electrons to achieve stability.
Group 14
This is carbons family, one of the most abundant elements which shapes all life as we know it. These elements will readily form compounds. They have a valency of four, which means when you react say carbon and hydrogen together you get methane – CH4. I actually think it is quite inspiring to look at carbon:

Now just think how this impacts everything and everyone around you. Here is a list of some carbon compounds;
- carbon dioxide (CO2)
- carbon monoxide (CO)
- chloroform (CHCl3)
- carbon tetrachloride (CCl4)
- methane (CH4)
- ethylene (C2H4)
- acetylene (C2H2)
- ethyl alcohol (C2H5OH)
- acetic acid (CH3COOH)
The list really could go on forever – but glucose if a very important emission from this list, which clearly is everything for humans – it is the petrol we run on. I hope you are getting the point with valency that all of these elements in the group have the same valency. So because I have told you what carbon does you can work down the group and consider other elements – the valency is the same.
Group 15-17
First is the nitrogen family – which is a fairly important family because nitrogen is the first sufficiently electronegative element to be able to participle in electron bonding. Next we have the oxygen family and finally the halogens. What to observe is that the top element in the three groups can participate in hydrogen bonding – this will become slightly clearer considering general trends.
Overall the valency of the group is 3,2,1 as we go across – hence indicating why chlorine could appear in the above as NaCl however we need CO2 in order to get the chemistry to work.
Another thing to note is that these groups are not metals – which makes them distinct from earlier groups.
Group 18
The Noble gases – the perfect atoms. I like the word noble for these, it’s so loaded with personification. You can just imagine these atoms with perfect electron configuration that have been awarded with noble status while the ordinary imperfect atoms run around trying to be like them… this is exactly what happens. The noble gasses have a full valency shell which shields them from participating in any chemical reactions.
This is not quite true – with a huge amount of energy you can add or remove electrons from a noble gas, however for all intents and purposes they are un-reactive.
General trends
You should also note the following trends when trying to understand the table:
- From left to right across a period of elements, electronegativity increases.
- From top to bottom down a group, electronegativity decreases.
- Important exceptions of the above rules include the noble gases, lanthanides, and actinides.
There are lots of other trends – too many to list out, but they will come in future posts as we examine the materials in more detail. For today, I just wanted to do a whistle stop tour of the table. I know I have dropped in a huge number of terms with no explanation – but do ask if you have a question. Let me know other cool trends I didn’t mention – I ran out of time!

One of my favourite subjects…sadly, I only had half of a school year of it in my senior year. And chemistry is not something you can TRULY learn about, simply by reading…it eventually becomes ‘hands on’.
Very interesting post; looking forward to ‘listening in’ on the comments which are sure to follow, and learning from them and future posts in this subject…as always!😉
Pearl
LikeLiked by 2 people
I can’t help feeling a bit sorry for the noble gases ….their impermeable shell could mean they’re missing out on some kind of hitherto unheard of yet amazing reaction:)
Of course I don’t know what I’m talking about …am still awaiting my books ….they’re on their way tho apparantly ….meanwhile I am enjoying your and Dragullar’s blogs immensely ….learning a little Hindi too …no …no …don’t laugh ….just loving the Bloggosphere …it’s a way to a free education:)
LikeLiked by 3 people
At least they know where they are, can you imagine being a group 1 metal? One day you’re sodium and things are fine and then BANG you’re salt. Mind bending.
LikeLiked by 1 person
I would not guess to meet you even here! Amazing how people here are connected! Keep learning 🙂
LikeLiked by 1 person
Hi there, what do you mean when you talk about valency?
I like your writing style, it flows well and that makes it easy to follow.
LikeLiked by 2 people
Hey Charlotte thank you so much for your comments :). The valence electrons are the electrons which reside in the outer shell.. these are the most important because they interact first and are involved in chemical reactions. The valency is like…. the number of little “hooks” an atom has and tells us how it will interact. Hydrogen is an easy example as it needs one electron to complete its shell so it has a valency of one… if something has a valency of 2, say oxygen then you get a formula H2O – water. It is basically the number of electrons they need to loose/gain… it will tell you a lot about the reaction and if there will be a single bond, double bond etc. Does that make sense?
LikeLike
Your explanation makes perfect sense Joseph, thank you. I enjoy your blog a lot, plenty of science communication content which is fantastic.
LikeLiked by 1 person
Thank you so much, it’s great of you to visit 🙂
LikeLike
Shells? Or clouds? Or maybe just probabilities of being somewhere….. or not!
LikeLiked by 1 person
Clouds is my favourite but really there is no concrete way of describing electrons without using the mathematical framework of quantum mechanics… that said, simpler descriptions give very deep insight
LikeLike
Being in the Fire Service, I’ve had many a run in with beautiful magnesium. I guess car manufacturers never thought about a car possibly being on fire and the use of water to extinguish it. All I can say is that its ooooo, aaahhh and deluge, boom and deluge some more. It’s all fun and games till you have an auto extrication. I’m loving your posts.
LikeLiked by 2 people
Wow that really is interesting… and a little scary too! Some of the few scenarios where water actually fuels the flames! Perhaps car manufacturers should all have a course with a chemist before they are allowed to build cars!
LikeLiked by 1 person
It’s primarily old models that carried it in the engine block. You can trust me when I say, while manufacturers have left magnesium behind, they’ve come up with new, inventive ways of making our job beyond difficult. Collaboration is always the best idea. 🙂
LikeLiked by 1 person
I totally agree! Whilst I do not know all that much about cars, that sure does seem like a terrible dangerous idea – to have such a reactive metal in a hot moving vehicle
LikeLiked by 1 person
Kinda makes one wonder who makes the decesions, doesn’t it? I’ve had a couple incidents where the explosion was so violent it blew my partner and I back. Extremely dangerous. It is beautiful though.
LikeLiked by 1 person
Most beautiful things are also fairly dangerous…. life!
LikeLiked by 1 person
I love chemistry, so I loved reading this post! Do your studies include more in depth information on transition metals? They are truly fascinating 🙂
LikeLiked by 2 people
They will indeed! You know I never even took A-Level chemistry, so going straight in at undergrad has been a little bit rough but totally fascinating
LikeLiked by 1 person
Hope you enjoy it!!
LikeLike
Thanks for the lesson and for visiting my site yesterday. Please visit often! —Kyle
LikeLiked by 2 people
And thank you for visiting mine. Keep on running! Joe
LikeLike
Hey Joseph,
Awesome to see someone combine science and rationality – two of my favourite topics. I can’t wait to hear your thoughts on topics that i have in the pipeline to publish. keep it up man.
LikeLiked by 2 people
Thank you so much for your visit!
LikeLike
Every year we have a chemistry contest called the sodium boat challenge. The aim is to make an origami boat out of a 5cmx5cm piece of paper, which can hold a piece of sodium for the longest amount of time.
LikeLiked by 2 people
That’s amazing! I love it
LikeLiked by 1 person
Ah, dear old Mendeleev an unsung hero, but one of mine since the 1960’s. His structural insight followed me from being a scientist into becoming a psychologist!
LikeLiked by 2 people
He is indeed a very interesting person to read about from a critical thinking perspective
LikeLike
Joseph if I keep reading your material I might just learn how little I know about science. O wait a minute, I already know that much, all kidding aside, you are a good read sir.
LikeLiked by 1 person
Thank you very much! And you will be pleased to know no matter how much or how little you know, it always feels like there is so much more to know in science
LikeLike
It just reminded me of a book ‘Disappearing spoon’, simple and easy to understand.
LikeLiked by 1 person
I have never heard of it I will check it out!
LikeLiked by 1 person
Hmmm just read this post through again ….you are absolutely bloody right chemistry should not be overlooked by any discipline …something needs to be done with the pesky Co2 conundrum ….neutralise? pop? blast? ….I don’t know …we’re looking to you guys to get your heads together on this one ……it’s no good spending all that money on one man popping off to enjoy the view from space …although space is fascinating …we need something sorting with the earths climate to at least prolong our existence long enough to organise the global party ….and as for those noble gases WELL it’s a bit like politics they ain’t half difficult to shift;)
And yes ..yes …still reading in fits and starts …get a bit carried away then grind to a halt …then it’s off again …but that’s how I seem to work:D:D:D
LikeLike
Hey, Joseph you like science deeply, that’s evident, but you also have the ability to bring science to everyone in an easy to follow language, much like Feynman and Greene. I am pleased to follow you. I am sure you will author a nice science book, or have you already?
LikeLiked by 1 person
Thank you very much! I am glad you have enjoyed reading, in fact communicating science is something I feel very strongly about and was the whole reason I st this site up… so I could have a go outside the confines of anything academic. I am no where near writing a book! I wish I was, perhaps one day. I really enjoyed you post about memory stores
LikeLiked by 1 person
That’s great! Your have written some really interesting articles.I will look forward to keep track of your posts. Thank you very much; and thanks for the compliments about my post!
LikeLike
https://www.youtube.com/channel/UCtqQD_csQHyo56ahQR6faZw
You should subscribe to my channel! I make chem easy and fun to learn 🙂
LikeLiked by 1 person
Thank you very much I shall check out later!
LikeLiked by 1 person